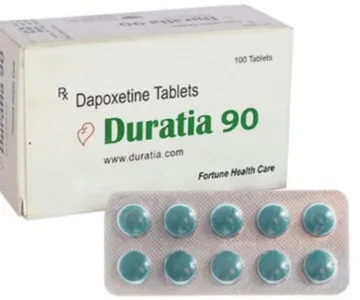
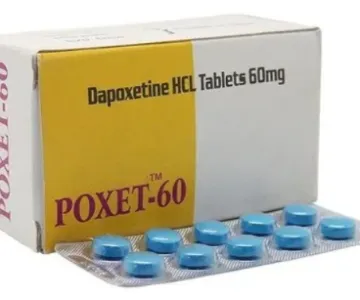
Priligy tablets are a drug recommended for premature ejaculation in adult men. Tablets are prescribed only to patients who have been diagnosed with PE. The main active ingredient, dapoxetine, is a potent selective serotonin reuptake inhibitor. Three dosages: 30 mg, 60 mg and 90 mg. Read more about the indications and features of the application in the instructions.
We sell generic Priligy under the following brand names: Duratia or Poxet. The price for one generic Priligy tablet starts from 2 USD.
Generic Priligy 30 mg:
| Package | Price | Per Pill | Savings | Savings |
|---|
Generic Priligy 60 mg:
| Package | Price | Per Pill | Savings | Savings |
|---|
Generic Priligy 90 mg:
| Package | Price | Per Pill | Savings | Savings |
|---|
Indications for use Priligy
Treatment of premature ejaculation (PE) in adult men aged 18 to 64 years.
Priligy is only recommended for use in patients who meet the following criteria:
- intravaginal ejaculation latency time (IELT) is less than two minutes;
- persistent or repeated ejaculation after minimal sexual stimulation before, during or shortly after sexual penetration, which occurs earlier than the moment desired by the patient;
- pronounced stress or difficulties that arose in interpersonal relationships as a result of PE;
- insufficient control over the onset of ejaculation;
- the onset of premature ejaculation in the majority of sexual intercourse attempts in the last 6 months.
Priligy should only be taken as a treatment, if necessary, before intended sexual intercourse.
Priligy should not be used to delay ejaculation in men who have not been diagnosed with PE.
Mode of application
The recommended starting dose for patients aged 18 to 64 years is 30 mg, which should be taken 1 to 3 hours before sexual intercourse. Treatment with Priligy should not be started with a dose of 60 mg.
Priligy® is not intended for continuous daily use. Priligy should only be taken if sexual intercourse is anticipated. The drug should not be taken more often than after 24 hours.
If the individual response to the drug at a dose of 30 mg is insufficient and the patient has not had moderate or severe adverse reactions, as well as prodromal symptoms suggesting the possibility of syncope, the dose can be increased to the maximum recommended dose of 60 mg, should be taken if necessary for about 1-3 hours before sexual intercourse. The frequency and severity of adverse reactions when taking the drug at a dose of 60 mg increases.
If the patient has experienced orthostatic reactions after taking the drug at the initial dose, it is not recommended to increase the dose to 60 mg. Do not exceed the maximum recommended dose.
A careful benefit-versus-risk analysis is needed when taking Priligy after the first four weeks of treatment (or at least after 6 doses of treatment) to determine whether it is reasonable to continue treatment with Priligy.
There are limited data on the efficacy and safety of using Priligy for more than 24 weeks. The clinical need for continued treatment and the risk-benefit ratio of Priligy treatment should be reassessed at least every six months.
Elderly patients (over 65 years old)
The efficacy and safety of Priligy has not been established in patients over 65 years of age.
Impaired kidney function
Caution should be exercised when taking the drug in patients with mild or moderate renal impairment. Priligy is not recommended for patients with severe renal impairment.
Impaired liver function
In patients with moderate or severe hepatic impairment, the drug is contraindicated.
Established slow metaboliser of CYP2D6 or patients treated with CYP2D6 inhibitors
Caution should be exercised in increasing the dose to 60 mg in patients with an established CYP2D6 slow metaboliser genotype or in patients who are concomitantly receiving potent CYP2D6 inhibitors.
Patients taking moderate-strength CYP3A4 inhibitors or strong CYP3A4 inhibitors
Concomitant use of potent inhibitors of CYP3A4 is contraindicated. Patients who are simultaneously taking moderate-strength CYP3A4 inhibitors should be careful not to exceed a dose of 30 mg.
Mode of application
The drug is intended for oral use. To avoid a bitter taste, tablets should be swallowed by breaking. Tablets should be taken with plenty of water. Priligy® can be taken with or without food.
Children
The use of Priligy is contraindicated in children due to lack of clinical experience.
Contraindications
- Hypersensitivity to the active substance or to any excipient of the drug.
- Heart failure (II-IV classes according to NYHA).
- Conduction disorders such as AV block or sick sinus syndrome.
- Severe ischemic heart disease.
- Severe valvular insufficiency.
- Having a history of fainting.
- A history of mania or severe depression.
- Simultaneous use of MAO inhibitors (MAO inhibitors) or if less than 14 days have passed after stopping their use. The use of Priligy® should be discontinued at least 7 days before the start of therapy with MAO inhibitors.
- Simultaneous administration of thioridazine or within 14 days after the end of administration. Priligy should be discontinued at least 7 days prior to initiation of thioridazine therapy.
- Concomitant use of serotonin reuptake inhibitors (selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs)) or other serotonergic drugs/herbal drugs [such as L-tryptophan, triptans, tramadol, linezolid, lithium, St. These medicines/herbal products should not be taken within 7 days after you stop taking Priligy.
- Concomitant administration of potent CYP3A4 inhibitors such as ketoconazole, itraconazole, ritonavir, saquinavir, telithromycin, nefazodone, nelfinavir, atazanavir and the like.
- Impaired liver function moderate or severe.
Compound
Active ingredient: Dapoxetine;
1 coated tablet contains dapoxetine hydrochloride 33.6 or 67.2 mg, which corresponds to dapoxetine 30 or 60 mg, respectively;
Excipients: lactose monohydrate, microcrystalline cellulose, croscarmellose sodium salt, anhydrous colloidal silicon dioxide, magnesium stearate.
Shell:
film-coated tablets, 30 mg: Powder Gray 4 (lactose, hypromellose, titanium dioxide (E 171), triacetin, iron oxide black (E172), iron oxide yellow (E172));
film-coated tablets, 60 mg: Powder Gray 3 (lactose, hypromellose, titanium dioxide (E 171), triacetin, iron oxide black (E172), iron oxide yellow (E 172).
Storage conditions
Special storage conditions are not required. Keep out of the reach of children.

Dear customers, be informed that all the information on this website was originally provided by the Ajanta Pharma Ltd. manufacturer of the Kamagra® remedy for men. Medical articles are reviewed by me. You may follow my biography.